While many signaling molecules affect dendrite development, the brain-derived neurotrophic factor (BDNF) and its immature predecessor proBDNF are particularly crucial in the central nervous system ( Lu et al., 2005). 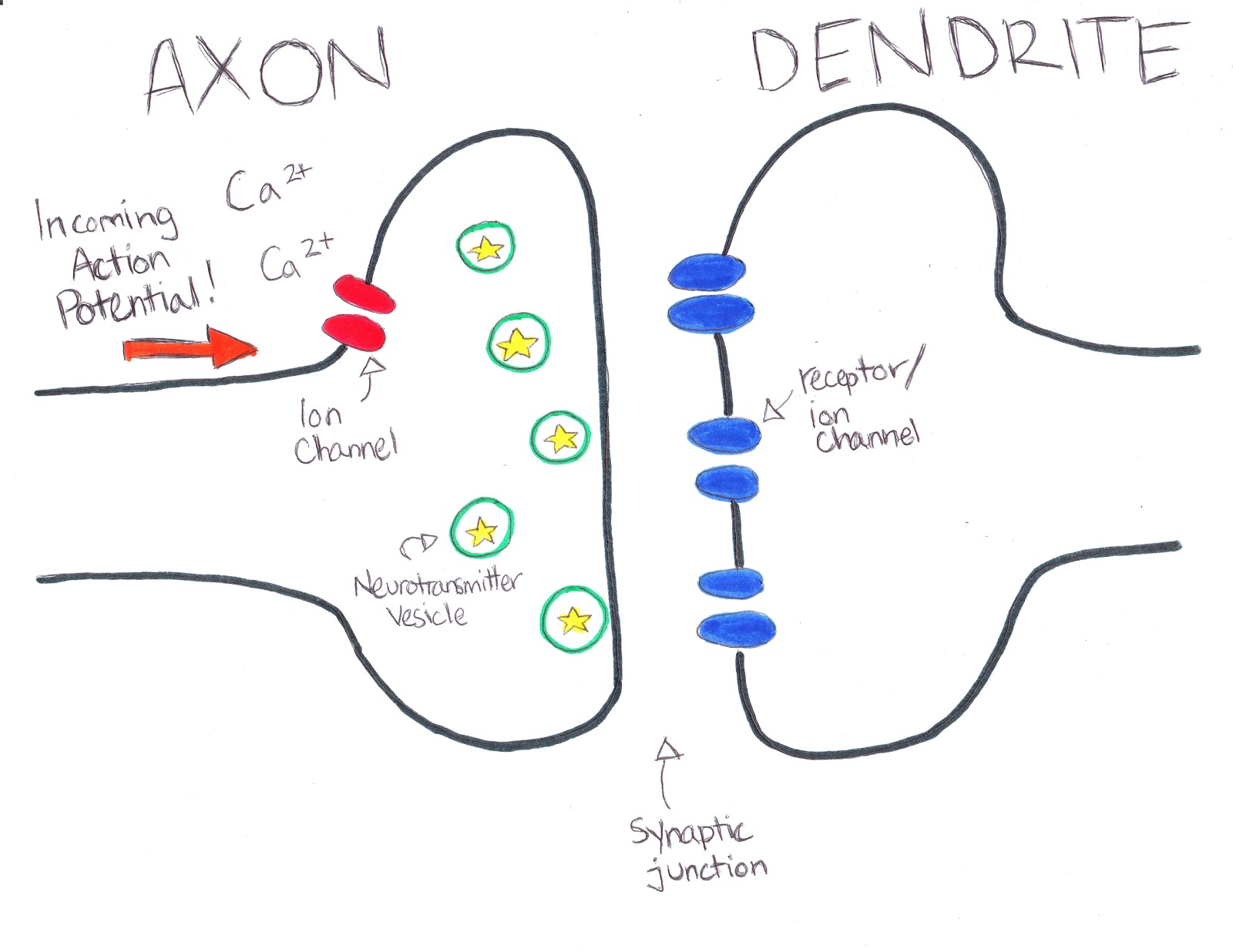
Extrinsic signaling, in contrast, exerts precise control over the detailed dynamics of dendrite development via various mechanisms, including activity-dependent cell-to-cell interactions and molecular signaling ( Polleux et al., 2016). In any given neuron, intrinsic expression of specific genes controls many morphological aspects, including the orientation of the dendrite in the cortex, the general abundance of dendritic branching, and the timing of growth onset ( Puram and Bonni, 2013). 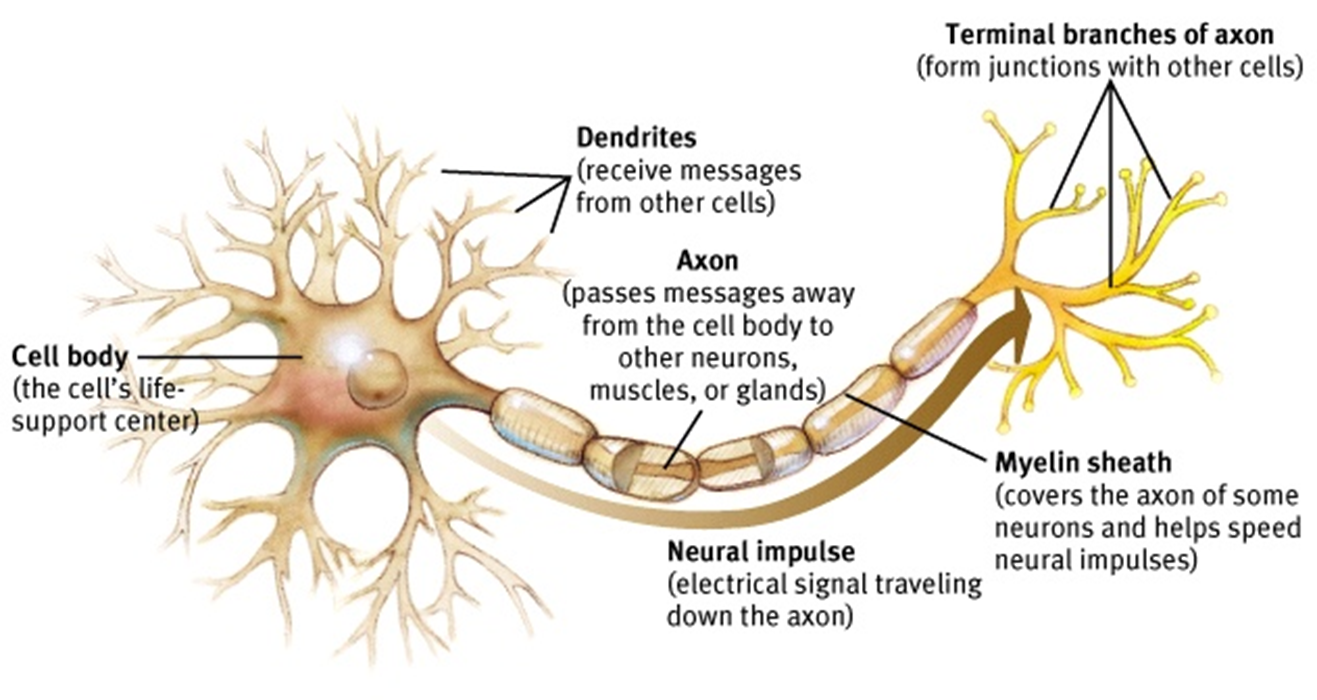
Many intrinsic and extrinsic factors underlie the dynamics of dendritic development. Dendrite development is highly dynamic iterated addition and retraction of branches allow these dendrites to probe various potential synaptic partners before stabilizing ( Cline, 2016 Richards et al., 2020). During development, the dendrites of many neurons grow simultaneously and become integrated into neural circuits. The morphology of dendrites determines the location of synaptic contacts with other neurons and thereby constrains single-neuron computations. 
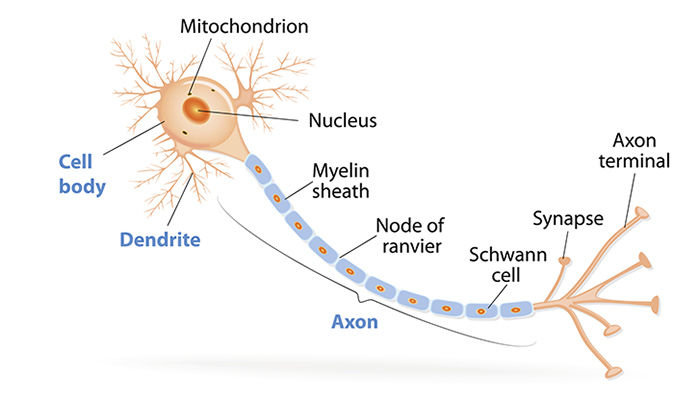
The dendrites of a neuron are intricately branched structures that receive electrochemical stimulation from other neurons. The work will be of interest to neuroscientists studying dendritic development and connectivity at a single-cell level. Some of the findings are a direct result of how the model is constructed, which limits the impact. The modelling is solid and could be broadly applied to other similar questions. This important work investigates how two distinct processes, morphological changes and synaptic plasticity, contribute to the final shape of neuronal dendrites and the spatial structure of their synaptic inputs. Our work suggests concrete mechanistic components underlying the emergence of dendritic morphologies and synaptic formation and removal in function and dysfunction, and provides experimentally testable predictions for the role of individual components. Therefore, a single mechanistic model can capture dendritic growth and account for the synaptic organization of correlated inputs during development. We demonstrate that a trade-off between activity-dependent and -independent factors influences dendritic growth and synaptic location throughout development, suggesting that early developmental variability can affect mature morphology and synaptic function. Besides setting up dendritic morphologies, activity-dependent plasticity rules organize synapses into spatial clusters according to the correlated activity they experience. Model-generated dendrites have approximately optimal wiring length consistent with experimental measurements. The model generates stellate-like dendritic morphologies capturing several morphological features of biological neurons under normal and perturbed learning rules, reflecting biological variability. Consistent with experiments, three phases of dendritic growth – overshoot, pruning, and stabilization – emerge naturally in the model. Here, we propose a mechanistic model in which dendrite growth and the organization of synapses arise from the interaction of activity-independent cues from potential synaptic partners and local activity-dependent synaptic plasticity. How they emerge simultaneously during brain development as neurons become integrated into functional networks is still not mechanistically understood. Dendritic branching and synaptic organization shape single neuron and network computations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
